
Nine ways to reduce liver abscess risk in beef on dairy cattle

Liver abscesses continue to be a challenge for the fed cattle industry, especially for the beef-on-dairy sector. They not only impact the health and well-being of the animal but may also result in substantial financial losses for cattle producers and beef packers, prompting producers to look for practical solutions. Implementing sound management and nutritional strategies on day one of the calf’s life can help reduce the prevalence of liver abscesses and their potential impact on cattle performance and operational profitability.
The primary bacterium found in cattle liver abscesses is Fusobacterium necrophorum. According to research conducted by Kansas State University Professor Dr. T.G. Nagaraja, DVM, this bacterium is a common denominator in liver abscess cases, even when other disease-causing organisms are present. While F. necrophorum is part of the normal gut microbiota and exposure to this bacterium is inevitable, whether it colonizes and flourishes in the animal’s body is a different matter.1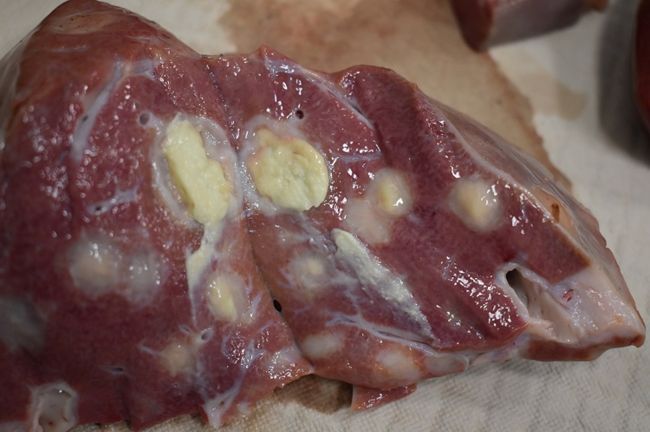
In dairy operations, feeding large volumes of colostrum via esophageal tube feeders can leave residual colostrum in the forestomachs, providing a nutrient-rich environment for bacteria like F. necrophorum to grow. This can lead to subclinical rumenitis, which often goes unnoticed. Poor management of calf starter feed can exacerbate this problem, which may increase gut permeability and predispose the liver to abscesses.
Good husbandry and proper feeding practices are essential to help reduce the incidence and severity of liver abscesses in beef-on-dairy calves. Consider the following management steps in cooperation with your herd nutritionist and local veterinarian to help mitigate the risks.
For more beef-on-dairy management strategies, visit purinamills.com/dairy-beef and view the Beef-on-Dairy Industry Report.
Reference:
1 Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University, Manhattan, Kansas 66506
The primary bacterium found in cattle liver abscesses is Fusobacterium necrophorum. According to research conducted by Kansas State University Professor Dr. T.G. Nagaraja, DVM, this bacterium is a common denominator in liver abscess cases, even when other disease-causing organisms are present. While F. necrophorum is part of the normal gut microbiota and exposure to this bacterium is inevitable, whether it colonizes and flourishes in the animal’s body is a different matter.1

Image of an abscessed bovine liver
Photo provided by the Wisconsin Veterinary Diagnostic Laboratory
In dairy operations, feeding large volumes of colostrum via esophageal tube feeders can leave residual colostrum in the forestomachs, providing a nutrient-rich environment for bacteria like F. necrophorum to grow. This can lead to subclinical rumenitis, which often goes unnoticed. Poor management of calf starter feed can exacerbate this problem, which may increase gut permeability and predispose the liver to abscesses.
Good husbandry and proper feeding practices are essential to help reduce the incidence and severity of liver abscesses in beef-on-dairy calves. Consider the following management steps in cooperation with your herd nutritionist and local veterinarian to help mitigate the risks.
- Feed smaller volumes of high-quality colostrum (IgG ≥75 g/L) to newborn calves. Feeding a smaller volume with the proper mass of IgG can allow for the successful transfer of passive immunity, and for calves fed colostrum via an esophageal tube feeder, it may reduce inflammation in the immature rumen.
- Feeding a high plane of nutrition or a minimum of 2.0 gallons of milk replacer and/or its equivalent in pasteurized milk per day helps avoid excessive hunger, promotes a gradual transition to starter feed and helps the rumen mature properly.
- Maintain a consistent feeding schedule with at least two feedings per day spaced 12 hours apart. Operations that feed calves within a short period, leaving them without a pasteurized waste milk or milk replacer meal for an extended period of time, can be predisposed to gut health issues.
- Slowly introduce a starter feed within the first few days. The starter ration should have limited fines and a proper balance of fiber and starch to support proper rumen maturation, along with adequate protein to foster muscle and gut tissue growth.
- Consider feeding a diet that includes direct-fed microbials, or probiotics that have been researched to show benefit in calves, to help protect the calf’s gut.
- Ensure fresh, clean, free-choice water is always available to encourage consistent water and starter intake. Do not provide soft water for drinking. Softened water can have a high sodium concentration, increasing the risk of sodium toxicosis. Test water-source quality at least twice per year.
- Feed milk replacer and/or pasteurized milk for a minimum of 8 weeks, with a preference of 10 weeks prior to starting the weaning process. This allows adequate time for rumen development.
- Provide a gradual two-week weaning process to ensure a smooth and healthy transition from a liquid diet to dry feed. Abrupt transitions can lead to gut inflammation, increased permeability and hindgut acidosis, which may negatively impact liver health.
- When moving calves into group pens, minimize stress by providing additional feed and water options during the first two weeks while calves acclimate to their new environment.
For more beef-on-dairy management strategies, visit purinamills.com/dairy-beef and view the Beef-on-Dairy Industry Report.
Reference:
1 Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University, Manhattan, Kansas 66506




